Unraveling an 80-Year-Old Mystery: Crystal Structure of a Bromide Hydrate Revealed by Synchrotron Radiation
Breakthrough in Understanding a Longstanding Scientific Mystery
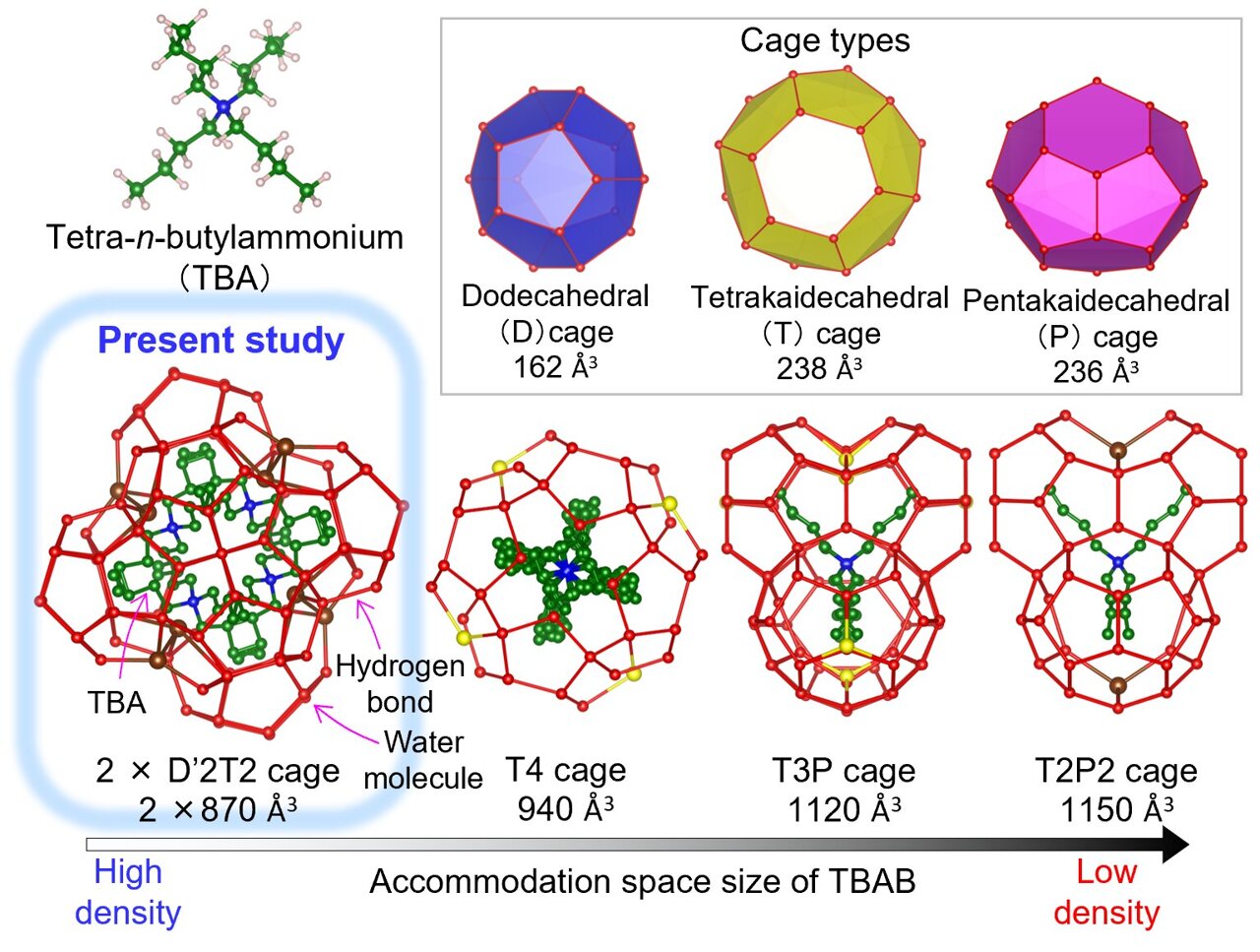
For over 80 years, scientists have struggled to determine the exact crystal structure of a compound known as tetra-n-butylammonium bromide hydrate (TBAB·26H₂O). This material is part of a group called semiclathrate hydrates, which are formed when ions combine with water. These materials have been widely used in various applications, including air conditioning systems. Now, researchers have finally uncovered the mystery and provided a clear understanding of its molecular arrangement.
The discovery was made possible through advanced scientific techniques and collaboration between researchers from different institutions. The findings were published in a recent paper titled Solving the 80-Year Structure Mystery: Definitive Crystal Structure of TBAB Hydrate Resolved with Synchrotron Radiation in the journal Crystal Growth & Design.
This breakthrough has significant implications for both scientific research and industrial applications. By understanding how TBAB·26H₂O forms and functions at the molecular level, scientists can develop more efficient materials for thermal energy storage and other practical uses. This could lead to improvements in energy efficiency and sustainability across multiple industries.
What Are Semiclathrate Hydrates?
Semiclathrate hydrates are a unique class of crystalline materials that consist of guest molecules trapped within a cage-like structure made of water molecules. These structures are held together by hydrogen bonds, which give them stability and specific physical properties. Water-based functional materials, such as hydrogels, aqueous polymers, and clathrate hydrates, are essential in many industrial processes due to their ability to interact with and store energy.
TBAB·26H₂O is a specific example of a semiclathrate hydrate that has been used extensively in thermal energy storage. It contains a guest molecule of tetra-n-butylammonium bromide (TBAB) surrounded by a network of water molecules. This structure allows it to store and release heat efficiently, making it ideal for use in air conditioning systems and other cooling technologies.
Despite its widespread use, the exact crystal structure of TBAB·26H₂O remained unknown for decades. Previous studies suggested a tetragonal lattice structure, but this did not fully explain all of the material’s properties. Researchers needed a more precise method to determine the actual arrangement of molecules within the crystal.
Using Synchrotron Radiation to Solve the Puzzle
To solve this long-standing mystery, scientists turned to synchrotron radiation, a powerful tool that provides high-energy X-rays capable of probing the atomic-level structure of materials. The research team used the Super Photon ring-8 (SPring-8) facility in Sayo Town, Japan, which is one of the most advanced synchrotron radiation sources in the world.
Through detailed analysis of the X-ray diffraction patterns, the researchers were able to determine that the crystal structure of TBAB·26H₂O is a tetragonal superstructure. This structure belongs to a known type of hydrate called Jeffrey's type III, but it has unique features that distinguish it from other similar compounds.
One of the key differences is the arrangement of the TBAB cation within the water cage. Unlike previous models, this new structure reveals a novel configuration that allows for a denser and more stable crystal lattice. This finding helps explain the material’s exceptional heat storage capabilities and opens up new possibilities for designing better hydrate-based materials.
Implications for Future Research and Applications
Understanding the crystal structure of TBAB·26H₂O is a major step forward in the development of sustainable and energy-efficient technologies. This knowledge can be applied to improve thermal storage systems, reduce energy consumption, and lower carbon emissions.
The researchers also plan to use this newfound insight to explore other hydrate-forming systems, including polymers and soft matter. By expanding these structural principles, they hope to create advanced materials that can be used in a variety of industrial applications, such as gas separation and carbon capture.
“This structure explains the material’s heat storage characteristics and provides new design principles for hydrate-based functional materials,” said Hironobu Machida, Chief Engineer at Panasonic Corporation. “Understanding this structure opens the door to engineering better thermal storage and related applications.”
Collaborative Efforts in Scientific Discovery
The study involved a team of researchers from several institutions, including Yokohama National University, the Japan Synchrotron Radiation Research Institute (JASRI), The University of Osaka, and Panasonic Corporation. Their combined expertise and access to cutting-edge facilities made this breakthrough possible.
As the field of hydrate science continues to evolve, this discovery serves as a foundation for future research and innovation. With a deeper understanding of these complex materials, scientists can work toward developing more sustainable and efficient solutions for global energy challenges.
Post a Comment for "Unraveling an 80-Year-Old Mystery: Crystal Structure of a Bromide Hydrate Revealed by Synchrotron Radiation"
Post a Comment