Rocket Stock Gains FDA RMAT for Heart Failure Gene Therapy
Rocket Pharmaceuticals Receives FDA Designation for Gene Therapy
Rocket Pharmaceuticals (RCKT) recently announced that the U.S. Food and Drug Administration (FDA) has granted the Regenerative Medicine Advanced Therapy (RMAT) designation to its investigational adeno-associated virus (AAV)-based gene therapy, RP-A601, for treating PKP2-arrhythmogenic cardiomyopathy (ACM). This designation is a significant milestone for the company as it aims to develop innovative treatments for rare genetic conditions.
PKP2-arrhythmogenic cardiomyopathy is a genetic heart condition caused by a defect in the PKP2 gene, which weakens the heart muscle and can lead to abnormal heart rhythms and an increased risk of sudden cardiac death. The RMAT designation offers several advantages, including increased opportunities for meetings with FDA officials and discussions on potential surrogate or intermediate endpoints. This designation also facilitates early interactions between the FDA and the sponsor, potentially leading to accelerated approval and priority review of the biologics license application (BLA).
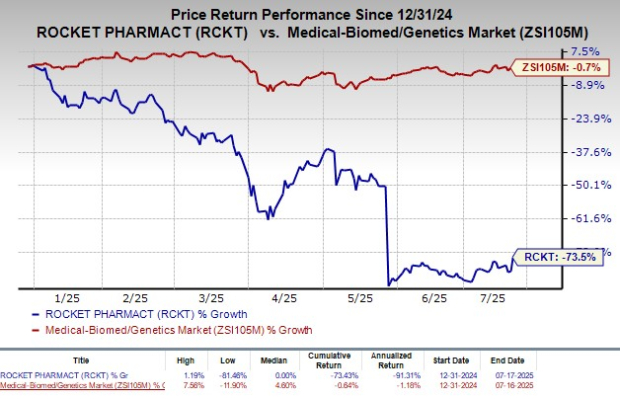
Following the announcement, RCKT's stock saw a significant jump, rising 16.8% on the day of the news. However, the stock has experienced a substantial decline this year, dropping 73.5%, compared to the industry’s modest decrease of 0.7%. This highlights the volatile nature of the biotech sector and the challenges companies face in navigating regulatory and market dynamics.
Positive Results from Phase I Study
The FDA’s RMAT designation was based on positive safety and efficacy data from the phase I study evaluating RP-A601 for PKP2-ACM. Preliminary results from the ongoing phase I study showed encouraging early safety and efficacy outcomes. Additionally, treatment with RP-A601 was generally safe and well-tolerated, indicating a promising trajectory for the therapy.
In addition to RP-A601, Rocket Pharmaceuticals is developing another AAV-based gene therapy candidate, RP-A701, for treating BAG3-associated dilated cardiomyopathy (BAG3-DCM), a rare and inherited heart condition. The FDA recently cleared the company’s investigational new drug application for RP-A701, and the company plans to conduct a phase I study evaluating RP-A701, an AAVrh.74-based gene therapy for the treatment of BAG3-DCM.
BAG3-DCM is a genetic heart disease that causes the heart muscles to become enlarged and weakened, ultimately leading to heart failure. The development of RP-A701 represents another step forward in Rocket Pharmaceuticals’ efforts to address rare and complex heart conditions through gene therapy.
Recent Challenges and Pipeline Focus
Despite these advancements, Rocket Pharmaceuticals has faced several setbacks in recent months. In May 2025, the company voluntarily paused dosing in the pivotal phase II study evaluating its gene therapy candidate, RP-A501, for treating patients with Danon disease, following a patient death. The FDA placed a clinical hold on the study, highlighting the risks associated with developing therapies for rare diseases.
In June 2024, the FDA issued a complete response letter to the BLA seeking approval for Kresladi to treat LAD-I, a rare genetic disorder. This marked the second time the FDA requested additional information from the company regarding Kresladi’s Chemistry Manufacturing and Controls, part of the BLA submission. In February 2024, the FDA extended the review period of the Kresladi BLA by an additional three months.
These challenges underscore the importance of continued focus on the successful development of other gene therapy candidates. Rocket Pharmaceuticals remains committed to advancing its pipeline and addressing unmet medical needs in the field of genetic disorders.
Zacks Investment Research Insights
Rocket Pharmaceuticals currently carries a Zacks Rank of #3 (Hold), reflecting a neutral outlook from analysts. While the company’s performance has been mixed, there are other biotech stocks with more favorable rankings. For instance, Arvinas (ARVN), Keros Therapeutics (KROS), and Akero Therapeutics (AKRO) each have a Zacks Rank of #1 (Strong Buy), indicating strong confidence in their future prospects.
Arvinas has seen improvements in earnings estimates over the past 60 days, with losses per share for 2025 narrowing from $1.60 to $1.51. Similarly, Keros Therapeutics has improved its loss per share estimates for 2025 from 79 cents to 29 cents. Akero Therapeutics has shown significant growth, with shares surging 95.3% year to date.
Each of these companies has demonstrated varying levels of earnings performance, with some beating estimates and others missing them. These insights provide a broader context for investors considering biotech stocks and highlight the diverse landscape of the sector.
Post a Comment for "Rocket Stock Gains FDA RMAT for Heart Failure Gene Therapy"
Post a Comment