FDA Panel Rejects GSK's Blenrep Combination Therapy
GSK Faces Regulatory Challenges with Blenrep Combo Therapy
GSK plc recently announced that the U.S. Food and Drug Administration’s (FDA) Oncologic Drugs Advisory Committee (ODAC) has voted against the overall benefit/risk profile of Blenrep (belantamab mafodotin-blmf) in combination therapy at the proposed dosing regimen. This decision suggests that the committee believes the potential risks associated with the treatment currently outweigh its benefits for adults with relapsed or refractory multiple myeloma.
Following this announcement, shares of GSK dropped by 4.7% on the day of the news. The ODAC’s recommendation comes just a week before the FDA is expected to make its final decision on the biologics license application (BLA) for Blenrep. The BLA sought approval of the drug in combination with J&J’s Velcade (bortezomib) plus dexamethasone (BorDex) or Bristol Myers’ Pomalyst (pomalidomide) plus dexamethasone (PomDex) for patients who have received at least one prior line of therapy.
Given that the FDA typically takes the ODAC’s opinion into consideration, it is likely that the regulatory body may reject the approval of the Blenrep combo in the United States. This outcome could significantly impact GSK’s efforts to revive the drug in the U.S. market.
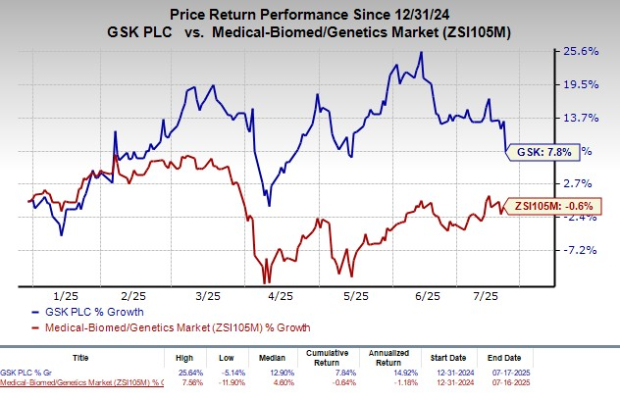
GSK's Stock Performance
Year to date, GSK’s stock has seen a rise of 7.8%, outperforming the broader industry, which experienced a slight decline of 0.6%. This positive performance highlights the company’s resilience despite recent setbacks.
Details on GSK’s Blenrep Combo
The BLA for the Blenrep combo was based on data from two phase III clinical trials, DREAMM-7 and DREAMM-8, both of which met their primary endpoints related to progression-free survival (PFS). However, regulatory applications for the combination therapy are currently under review in the European Union and other regions.
It is important to note that in November 2022, Blenrep was withdrawn from the U.S. market after the failure of GSK’s phase III confirmatory study, DREAMM-3. This trial did not meet its primary endpoint of PFS, which led to the withdrawal of the drug under the FDA’s accelerated approval regulations. As a result, Blenrep monotherapy was also removed from the EU market.
Despite these challenges, Blenrep combinations were approved in the United Kingdom and Japan in April and May 2025, respectively. However, at present, there is little likelihood of the Blenrep combo receiving U.S. approval, which poses a setback for GSK’s plans to reintroduce the drug in the country.
Zacks Rank and Biotech Stocks to Watch
Currently, GSK holds a Zacks Rank of #3, indicating a "Hold" rating. Investors interested in biotechnology stocks may consider alternatives such as:
- Arvinas (ARVN): With a Zacks Rank of #1, this stock has shown strong performance, though it has declined year-to-date by 59.5%.
- Keros Therapeutics (KROS): Holding a Zacks Rank of #1, KROS has decreased by 9.3% year-to-date.
- Akero Therapeutics (AKRO): Also ranked #1, AKRO has surged by 95.3% year-to-date.
Each of these companies has demonstrated varying levels of earnings performance, with some beating estimates consistently and others missing them in certain quarters. These fluctuations highlight the volatility and potential of the biotech sector.
Investors looking for more insights into top-performing stocks can explore the complete list of today’s Zacks #1 Rank stocks.
Post a Comment for "FDA Panel Rejects GSK's Blenrep Combination Therapy"
Post a Comment